The table below shows some physical properties of two unkown samples
Physical Properties:
Sam...

Chemistry, 01.12.2021 20:40 kakesheco4210
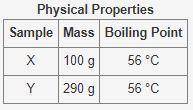
The table below shows some physical properties of two unkown samples
Physical Properties:
Sample 1: X, Mass: 100g, Boiling Point: 56 °C
Sample 2: Y, Mass: 290g, Boiling Point: 56 °C
What answer would best support whether the two samples are of the same substance or not?
A. The two substances are the same because their extensive property is different.
B. The two substances are not the same because their extensive property is the same.
C. The two substances are the same because their intensive property is the same.
D. The two substances are not the same because their intensive property is different.
An image if needed:

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1


Chemistry, 22.06.2019 12:00
Give the set of reactants (including an alkyl halide and a nucleophile) that could be used to synthesize the following ether: draw the molecules on the canvas by choosing buttons from the tools (for bonds and charges), atoms, and templates toolbars, including charges where needed. ch3ch2och2ch2chch3 | ch3
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
Questions

Mathematics, 09.06.2021 05:40


Social Studies, 09.06.2021 05:40

Chemistry, 09.06.2021 05:40

Mathematics, 09.06.2021 05:40

Biology, 09.06.2021 05:40

Biology, 09.06.2021 05:40

Biology, 09.06.2021 05:40

Biology, 09.06.2021 05:40





Business, 09.06.2021 05:40

Mathematics, 09.06.2021 05:40

English, 09.06.2021 05:40






