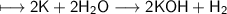Chemistry, 28.11.2021 09:50 sterlingrobinson35
When potassium metal is placed in water, a large amount of energy is released as potassium hydroxide and hydrogen gas are produced in the reaction 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g). Your lab partner says this is a redox reaction and a combustion reaction. Do you agree? Defend your answer by explaining whether or not it meets the requirements of each type of reaction.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Complete this brønsted-lowry reaction placing each product by its appropriate label. hso4- + hcn
Answers: 1

Chemistry, 22.06.2019 05:10
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2

Chemistry, 23.06.2019 01:30
How does the attraction between particles affect the ability of a solvent to dissolve in a substance
Answers: 1
You know the right answer?
When potassium metal is placed in water, a large amount of energy is released as potassium hydroxide...
Questions



Mathematics, 09.01.2021 03:00

Mathematics, 09.01.2021 03:00




Mathematics, 09.01.2021 03:00


Chemistry, 09.01.2021 03:00



Biology, 09.01.2021 03:00

History, 09.01.2021 03:00


Biology, 09.01.2021 03:10


Mathematics, 09.01.2021 03:10


Mathematics, 09.01.2021 03:10