I have a mixture of three gases in a flask at 25 C. The total pressure in the flask is 26.2 psi. If my gas mixture consists of 13.2 g of CHCl3, 25.8 g of HCl, and 3.2 g of H2, what is the pressure of each gas in the flask? Give each pressure to the nearest 0.1 psi:

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 23:30
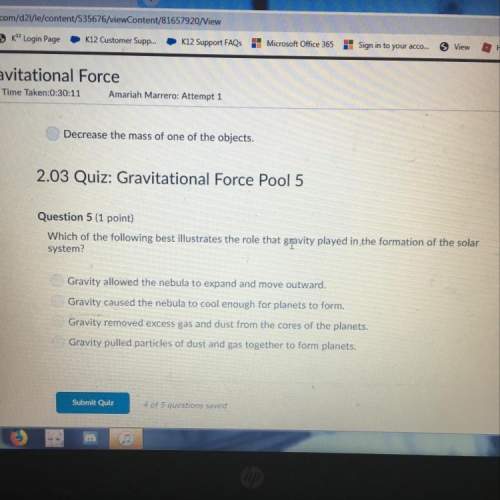
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1


Chemistry, 23.06.2019 03:10
Which of the following compounds would be expected to have the strongest ionic bonds? a)the compound that has b)the largest ions with the greatest charge c)the compound that has d)the largest ions with the least charge the compound that has the smallest ions with the greatest charge the compound that has the smallest ions with the least charge
Answers: 2
You know the right answer?
I have a mixture of three gases in a flask at 25 C. The total pressure in the flask is 26.2 psi. If...
Questions


Mathematics, 01.04.2020 14:45



Biology, 01.04.2020 14:47

Social Studies, 01.04.2020 14:47



Mathematics, 01.04.2020 14:49

Mathematics, 01.04.2020 14:49






World Languages, 01.04.2020 14:51

English, 01.04.2020 14:51



History, 01.04.2020 14:54