
Chemistry, 28.10.2021 01:00 naiomireyes74p2aybs
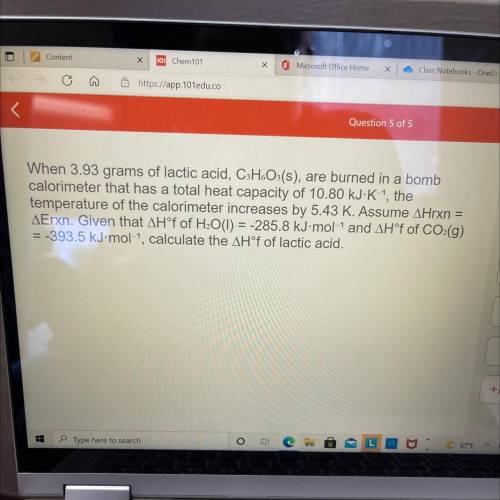
When 3.93 grams of lactic acid, CHoOs(s), are burned in a bomb
calorimeter that has a total heat capacity of 10.80 kJ-K-1, the
temperature of the calorimeter increases by 5.43 K. Assume AHrxn
=
AErxn. Given that AH°f of H¿O(I) = -285.8 kJ-mol-1 and AH°f of CO:(g)
= -393.5 kJ• mol-1, calculate the AH°f of lactic acid.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 04:00
Electric charge is what ? a. kinetic energy b. radiation c. discovery d. electricity
Answers: 1

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
When 3.93 grams of lactic acid, CHoOs(s), are burned in a bomb
calorimeter that has a total heat c...
Questions




History, 18.10.2019 14:30


Health, 18.10.2019 14:30


History, 18.10.2019 14:30

Mathematics, 18.10.2019 14:30

Social Studies, 18.10.2019 14:50

English, 18.10.2019 14:50

Mathematics, 18.10.2019 14:50

Mathematics, 18.10.2019 14:50

English, 18.10.2019 14:50


Computers and Technology, 18.10.2019 14:50



History, 18.10.2019 14:50

Mathematics, 18.10.2019 14:50



