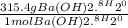WILL GIVE BRAINLIEST AND 20 POINTS! Barium hydroxide, often used to titrate weak organic acids, is obtained as the octahydrate, Ba(OH)2 * 8 H2O. What mass of Ba(OH)2 * 8 H2O would be required to make 500 mL of a solution that is 0.1500 M hydroxide ions? [hint: calculate the molar mass of barium hydroxide octahydrate].

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2

Chemistry, 23.06.2019 00:00
How is the way a mixture is combined different from how a compound is combined?
Answers: 3
You know the right answer?
WILL GIVE BRAINLIEST AND 20 POINTS!
Barium hydroxide, often used to titrate weak organic acids, is...
Questions


Mathematics, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10


Geography, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10

Business, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10


Mathematics, 04.03.2021 18:10

Mathematics, 04.03.2021 18:10


Mathematics, 04.03.2021 18:10


Arts, 04.03.2021 18:10


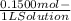 ×
×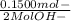 ×
×