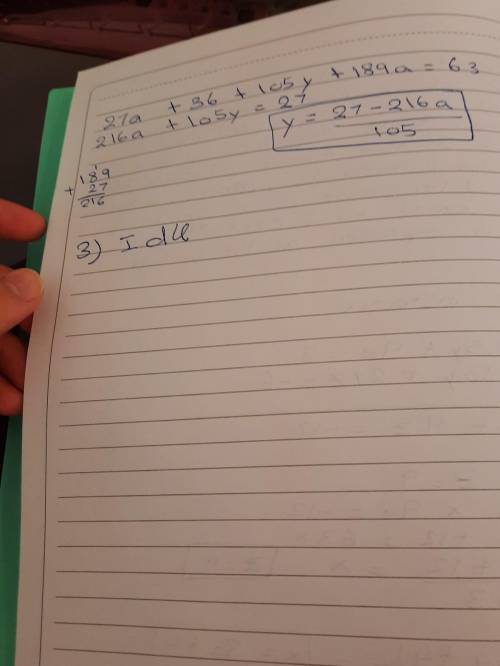1-Link the concepts of mole, molar mass, number of atoms, Avogadro’s
number and mass using a flowchart or mind map.
2-Rearrange the two equations to make each variable the focus (you will end up with three variations for each equation).
3- Record the equations for number of moles and number of particles (and
each equation triangle) - Annotate each symbol with what it means and definition and the units.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 22.06.2019 22:00
11) burning your hand when accidentally touching a hot plate is an example of which heat transfer? a. conduction b. convection c. radiation d. none of these
Answers: 2

Chemistry, 23.06.2019 03:00
Use the half-reactions of the reaction au(oh)3 + hi -> au +i2 +h2o to answer the questions
Answers: 1
You know the right answer?
1-Link the concepts of mole, molar mass, number of atoms, Avogadro’s
number and mass using a flowc...
Questions


Mathematics, 17.09.2021 15:50



Mathematics, 17.09.2021 15:50

Mathematics, 17.09.2021 15:50

Law, 17.09.2021 15:50




History, 17.09.2021 15:50

Mathematics, 17.09.2021 15:50

Mathematics, 17.09.2021 15:50



Mathematics, 17.09.2021 15:50



Mathematics, 17.09.2021 15:50