
Chemistry, 10.10.2021 06:20 dbn4everloved8
Properties and Changes Quiz (21-22) (COPY) / 4 of 14
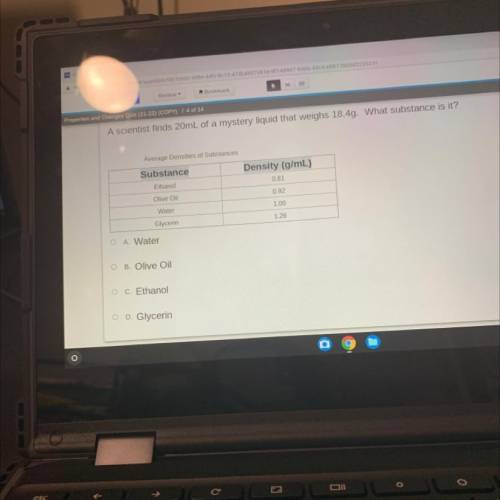
A scientist finds 20mL of a mystery liquid that weighs 18.4g. What substance is it?
Average Densities of Substances
Substance
Density (g/mL)
0.81
Ethanol
0.92
Olive Oil
Water
1.00
1.26
Glycerin
O A. Water
OB. Olive Oil
O C. Ethanol
O D. Glycerin

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 03:30
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
You know the right answer?
Properties and Changes Quiz (21-22) (COPY) / 4 of 14
A scientist finds 20mL of a mystery liquid th...
Questions



Physics, 18.11.2019 20:31


Mathematics, 18.11.2019 20:31

Arts, 18.11.2019 20:31





Mathematics, 18.11.2019 20:31

English, 18.11.2019 20:31

Chemistry, 18.11.2019 20:31

Mathematics, 18.11.2019 20:31

History, 18.11.2019 20:31


Mathematics, 18.11.2019 20:31

English, 18.11.2019 20:31

Mathematics, 18.11.2019 20:31



