Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2


Chemistry, 22.06.2019 06:00
What type of electromagnetic radiation has a shorter wavelength than blue light
Answers: 2

Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1
You know the right answer?
Х
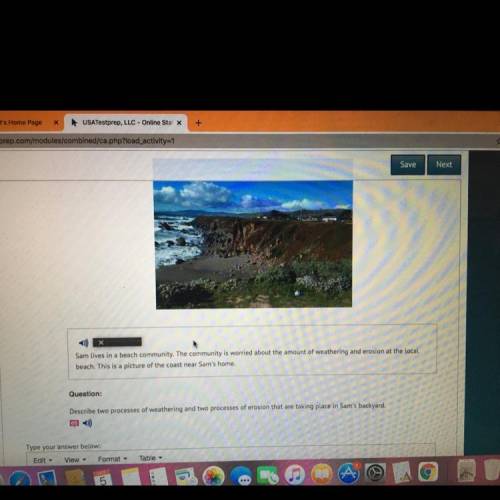
Sam lives in a beach community. The community is worried about the amount of weathering and eros...
Questions

History, 06.11.2019 09:31

English, 06.11.2019 09:31

Physics, 06.11.2019 09:31

Mathematics, 06.11.2019 09:31


Chemistry, 06.11.2019 09:31



Physics, 06.11.2019 09:31


History, 06.11.2019 09:31





Mathematics, 06.11.2019 09:31

English, 06.11.2019 09:31


Health, 06.11.2019 09:31