
Chemistry, 24.09.2021 01:00 ob3ymysins
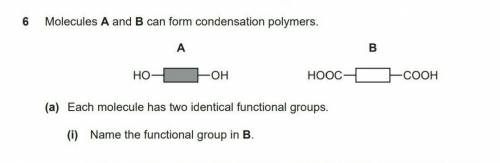
Why is the answer for this question 'carboxylic acid' and not 'carboxyl' Please anyone help! I will mark brainlliest to the best answer!

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1

Chemistry, 22.06.2019 18:00
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3

You know the right answer?
Why is the answer for this question 'carboxylic acid' and not 'carboxyl'
Please anyone help! I wil...
Questions

Mathematics, 29.03.2021 20:10


English, 29.03.2021 20:10

Computers and Technology, 29.03.2021 20:10



Mathematics, 29.03.2021 20:10



Social Studies, 29.03.2021 20:10


Mathematics, 29.03.2021 20:10


Mathematics, 29.03.2021 20:10

Mathematics, 29.03.2021 20:10

Spanish, 29.03.2021 20:10


Computers and Technology, 29.03.2021 20:10




