
Chemistry, 15.09.2021 15:40 artiomtyler007
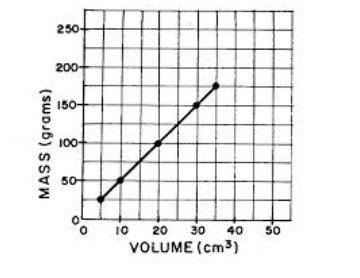
Can you unhelpful people answer this question already i'll mark you the brainlest. I'll mark you the brainlest if you answer the question. What is the independent variable shown on this graph
A. only volume (cm³) is the independent variable
B. both volume (cm³) and mass (grams) are independent variable
C. not enough information
D. the slope of the trend line
E. only mass (grams) is the independent variable

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2
You know the right answer?
Can you unhelpful people answer this question already i'll mark you the brainlest. I'll mark you the...
Questions


Mathematics, 03.04.2020 00:08





Mathematics, 03.04.2020 00:09

Mathematics, 03.04.2020 00:09

Mathematics, 03.04.2020 00:09



Physics, 03.04.2020 00:09

History, 03.04.2020 00:09

Spanish, 03.04.2020 00:09

Biology, 03.04.2020 00:09


Social Studies, 03.04.2020 00:10



History, 03.04.2020 00:10



