
Chemistry, 07.09.2021 23:40 wwwclarajohnson39
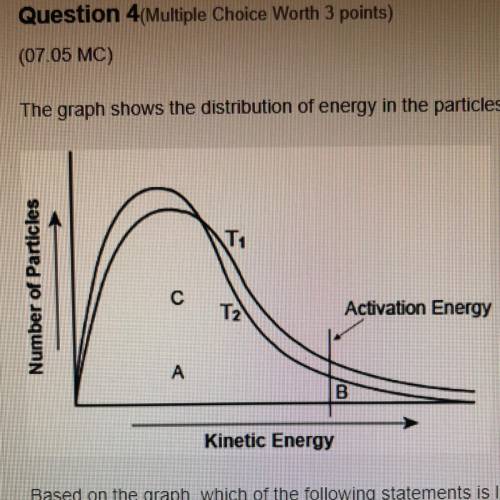
The graph shows the distribution of energy in the particles of two gas samples at different temperatures, T and T2. A, B, and C represent individual particles
Based on the graph, which of the following statements is likely to be true?
Particle A and C are more likely to participate in the reaction than particle B.
Most of the particles of the two gases have very high speeds.
A fewer number of particles of gas at T, are likely to participate in the reaction than the gas at T2
The average speed of gas particles at T2 is lower than the average speed of gas particles at T1

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:30
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 13:30
What does the xylem do? stores the glucose captures the sunlight absorbs oxygen into the leaf carries water from the roots to the leaves
Answers: 1

Chemistry, 23.06.2019 05:30
Find the midpoint of a segment with endpoints of 4-3i and -2+7i
Answers: 2
You know the right answer?
The graph shows the distribution of energy in the particles of two gas samples at different temperat...
Questions








Chemistry, 14.10.2019 17:30


History, 14.10.2019 17:30



History, 14.10.2019 17:30



English, 14.10.2019 17:30

World Languages, 14.10.2019 17:30

Health, 14.10.2019 17:30

Mathematics, 14.10.2019 17:30

Health, 14.10.2019 17:30




