3. Based on the following balanced equation:
2 C H20 + 13 02 8 CO2 + 10 H2O
a. How many moles...

Chemistry, 15.08.2021 01:00 lorraneb31
3. Based on the following balanced equation:
2 C H20 + 13 02 8 CO2 + 10 H2O
a. How many moles of O2 are required to react completely with 4.00 moles of C4H10?
3.00 6.00 8.00 10.00 13.00 16.00 20,00 23,00 26.00
b. How many moles of CO2 are produced after complete reaction of 4.00 moles of C H10?
3.00 6.00 8.00 10.00 13,00 16.00 20.00 23.00 26.00
c. How many moles of H2O are produced after complete reaction of 4.00 moles of C&H10?
3.00 6.00 8.00 10.00 13.00 1.6.00 20.00 23,00 26.00

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
Which term describes a fracture in the earth at which land stays in the same place? a. joint b. fault c. split d. hinge
Answers: 1

Chemistry, 21.06.2019 22:00
If 1.63 times 10 negative 4 of helium dissolves in 100.0g of water, what is the concentration in parts per million
Answers: 3

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1
You know the right answer?
Questions

Mathematics, 10.09.2019 06:10

Mathematics, 10.09.2019 06:10





Spanish, 10.09.2019 06:10



Mathematics, 10.09.2019 06:10

History, 10.09.2019 06:10



Mathematics, 10.09.2019 06:10


Chemistry, 10.09.2019 06:10

History, 10.09.2019 06:10


Chemistry, 10.09.2019 06:10

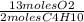 = 26 moles O2
= 26 moles O2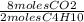 = 16 moles CO2
= 16 moles CO2 = 20 moles H20
= 20 moles H20

