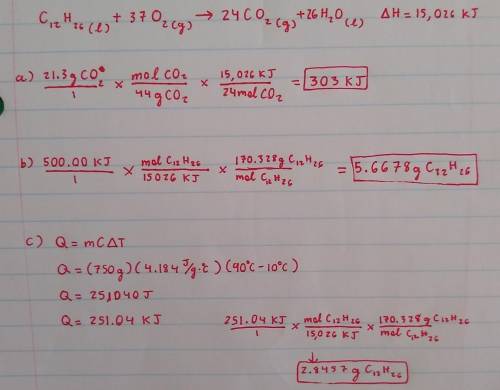Chemistry, 10.08.2021 07:10 yourmumsanoodle
1. Consider the following thermochemical reaction for kerosene:
2 C12H26(l) + 37 O2(g) 24 CO2(g) + 26 H2O(l) + 15,026 kJ
(a) When 21.3 g of CO2 are made, how much heat is released?
(b) If 500.00 kJ of heat are released by the reaction, how grams of C12H26 must have been consumed ?
(c) If this reaction were being used to generate heat, how many grams of C12H26 would have to be reacted to generate
enough heat to raise the temperature of 750g of liquid water from 10oC to 90oC?
2. Consider the reaction: NaNO3(s) + H2SO4(l) → NaHSO4(s) + HNO3(g) ΔH° = 21.2 kJ
How much heat must absorbed by the reaction system to convert 100g of NaNO3 into NaHSO4(s)?
3. What is the enthalpy change when 49.4 mL of 0.430 M sulfuric acid reacts with 23.3 mL of 0.309 M potassium
hydroxide?
3.
H2SO4(aq) + 2KOH(aq) → K2SO4(aq) + 2H2O(l) ΔH° = –111.6 kJ/mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

Chemistry, 23.06.2019 00:00
How is the way a mixture is combined different from how a compound is combined?
Answers: 3

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3
You know the right answer?
1. Consider the following thermochemical reaction for kerosene:
2 C12H26(l) + 37 O2(g) 24 CO2(g)...
Questions

Advanced Placement (AP), 02.11.2021 23:30


Mathematics, 02.11.2021 23:30

English, 02.11.2021 23:30

Chemistry, 02.11.2021 23:30


Chemistry, 02.11.2021 23:30

Mathematics, 02.11.2021 23:30

Mathematics, 02.11.2021 23:30

Mathematics, 02.11.2021 23:30



Mathematics, 02.11.2021 23:30

English, 02.11.2021 23:30



Advanced Placement (AP), 02.11.2021 23:30

English, 02.11.2021 23:30