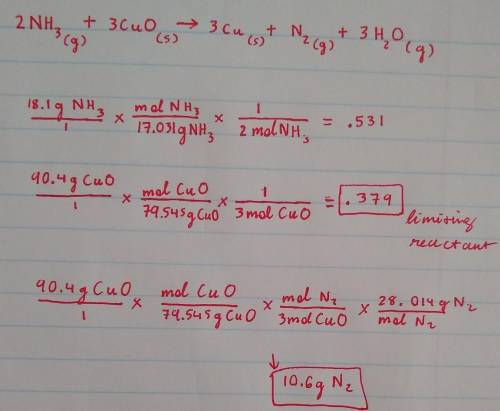Chemistry, 07.08.2021 14:00 zanedog2018
Nitrogen gas can be prepared by passing gaseous ammonia over solid copper (II) oxide at high temperatures. If 18.1 g of Nh3 is reacted with 90.4 g of CuO, which is the limiting reactant? How many grams of N2 will be formed? Explain how you solved for your answers. Balanced Equation: 2NH3 + 3CuO → 3Cu + N2 + 3H2O

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 15:30
A1.5l container holds p.50 grams of an unknown gas at a pressure of 0.44 atm and a temperature of 50.c what is the molar mass of the unknown gas
Answers: 1

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 23:30
The sum of the oxidation numbers in a neutral compound is always
Answers: 2
You know the right answer?
Nitrogen gas can be prepared by passing gaseous ammonia over solid copper (II) oxide at high tempera...
Questions


Mathematics, 20.08.2021 21:00




Mathematics, 20.08.2021 21:00




Mathematics, 20.08.2021 21:00

Mathematics, 20.08.2021 21:00


Computers and Technology, 20.08.2021 21:00



Mathematics, 20.08.2021 21:00

Mathematics, 20.08.2021 21:00

Mathematics, 20.08.2021 21:00


Health, 20.08.2021 21:00