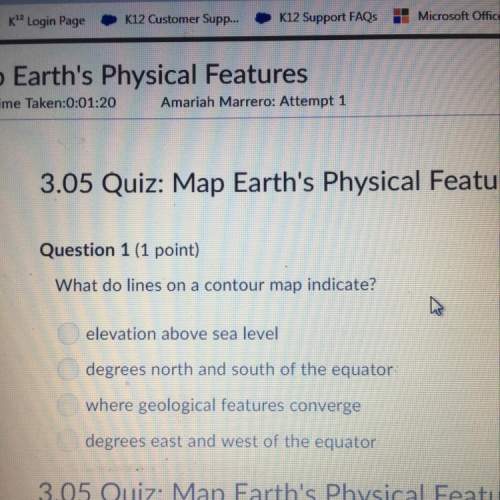
Chemistry, 06.08.2021 01:00 steventhecool22
If 14.0 grams of C2H4 and 155 grams of O2 are reacted in the balanced equation below and 38.0 grams of CO2 is collected, calculate the percent yield of carbon dioxide ? C2H4 + 3 O2 2 CO2 + 2 H2O

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
List four observations that indicate that a chemical reaction may be taking place
Answers: 1



Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1
You know the right answer?
If 14.0 grams of C2H4 and 155 grams of O2 are reacted in the balanced equation below and 38.0 grams...
Questions




Mathematics, 03.03.2021 14:00







Chemistry, 03.03.2021 14:00




English, 03.03.2021 14:00


Mathematics, 03.03.2021 14:00

English, 03.03.2021 14:00

Biology, 03.03.2021 14:00