
Chemistry, 30.07.2021 14:20 ddddre3909
According to the Vaporization Heat table, the heat needed for 1 mol of H2O to evaporate at 100°C is 40.7KJ and 44.0KJ/mol is needed to evaporate H2O at 25°C. Thus 44.0-40.7=3.7KJ is the energy needed to heat H2O to 100°C from 25°C.
However, according to the heat capacity of H2O, 3.7KJ will only warm the water by ~+43°C, which is not enough to reach 100°C starting from 25°C!
Am I missing something?!
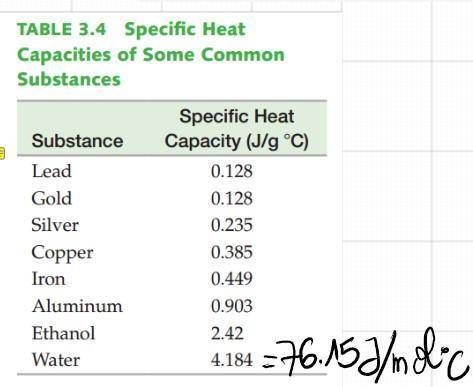
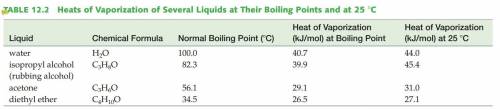

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 12:00
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1

Chemistry, 22.06.2019 17:00
Complete each row of the table below by filling in the missing prefix or missing exponent.
Answers: 1
You know the right answer?
According to the Vaporization Heat table, the heat needed for 1 mol of H2O to evaporate at 100°C is...
Questions


Mathematics, 01.12.2021 02:20

Mathematics, 01.12.2021 02:20


Mathematics, 01.12.2021 02:20


Computers and Technology, 01.12.2021 02:20

Mathematics, 01.12.2021 02:20


Geography, 01.12.2021 02:20

Mathematics, 01.12.2021 02:20





Mathematics, 01.12.2021 02:20



Mathematics, 01.12.2021 02:20




