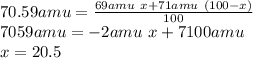Chemistry, 29.07.2021 14:00 asianaenaeh
Que. 1. Gallium has 2 naturally occuring Isotopes with mass numbers 69 & 71 respectively. What is the percentage abundance of both Isotopes if their relative atomic mass is equal to 70.59 Amu (Atomic mass Unit)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:10
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 17:00
Which statement is true about a catalyst? a: a catalyst decreases the rate of the reaction. b. a catalyst is consumed during a chemical reaction. c. a catalyst lowers the activation energy of a reaction. d. a catalyst increases the reactant concentration during a reaction.
Answers: 1

You know the right answer?
Que. 1. Gallium has 2 naturally occuring Isotopes with mass numbers 69 & 71 respectively. What i...
Questions





Biology, 01.04.2021 08:30

Advanced Placement (AP), 01.04.2021 08:30

Biology, 01.04.2021 08:30

Mathematics, 01.04.2021 08:30





Social Studies, 01.04.2021 08:30


History, 01.04.2021 08:30

History, 01.04.2021 08:30

Biology, 01.04.2021 08:30

Chemistry, 01.04.2021 08:40

English, 01.04.2021 08:40

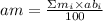
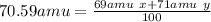 [1]
[1]