Chemistry, 28.07.2021 23:10 ldpozorski
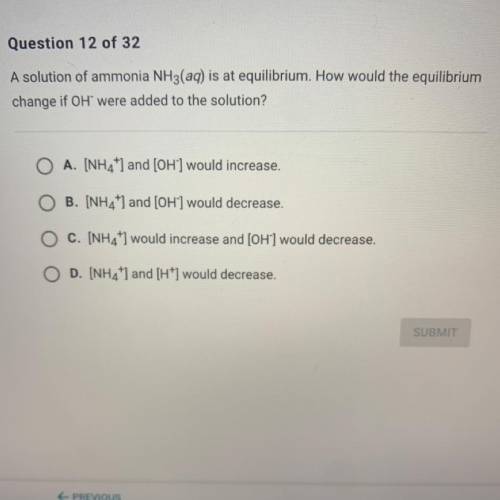
A solution of ammonia NH3(aq) is at equilibrium. How would the equilibrium
change if OH were added to the solution?
A. [NH4+] and [OH-] would increase.
B. [NH4+] and [OH-] would decrease.
C. [NH4+] would increase and [OH-] would decrease.
D. [NH4+] and [H+] would decrease.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
You know the right answer?
A solution of ammonia NH3(aq) is at equilibrium. How would the equilibrium
change if OH were added...
Questions

Physics, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

History, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01


Mathematics, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

Biology, 02.10.2020 09:01


English, 02.10.2020 09:01

Chemistry, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

English, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01


English, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01