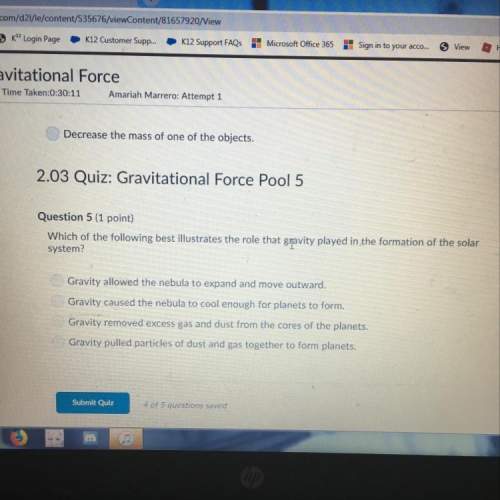
A system fitted with a piston expands when it absorbs 53.1 ) of heat from the surroundings. The piston is working against a pressure of 0.677 atm. The final volume is 63.2 L. What was the initial volume of the system if the internal energy of the system decreased by 108.3 J?
a. 65.6 L
b. 64.0 L
c. 70.8 L
d. 60.8 L
e. 54.4L

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 19:00
How many liters of ethylene glycol antifreeze (c2h6o2), with a density of 1.100 g/l, would you add to your car radiator containing 15.0 kg of water if you needed to protect your engine to - 21.5°c? for water, kf = 1.86°c m -1.
Answers: 1

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1
You know the right answer?
A system fitted with a piston expands when it absorbs 53.1 ) of heat from the surroundings. The pist...
Questions


Computers and Technology, 31.12.2020 18:10



Arts, 31.12.2020 18:10

Arts, 31.12.2020 18:10

Mathematics, 31.12.2020 18:10

Mathematics, 31.12.2020 18:10

Social Studies, 31.12.2020 18:10

Mathematics, 31.12.2020 18:10










Mathematics, 31.12.2020 18:20