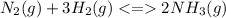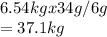Chemistry, 27.07.2021 02:00 dbzrules02
A reaction vessel for synthesizing ammonia by reacting nitrogen and hydrogen is charged with 6.54 kg of H2 and excess N2. A total of 30.4 kg of NH3 are produced. What is the percent yield of the reaction

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
10. translate each of the following chemical equations into a sentence. a. 2 zns(s) + 3 o2(g) -> 2 zno(s) + 2 so2(g) b. cah2(s) + 2 h2o(l) -> ca(oh)2 (aq) + 2 h2(g)
Answers: 2

Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3

You know the right answer?
A reaction vessel for synthesizing ammonia by reacting nitrogen and hydrogen is charged with 6.54 kg...
Questions





Social Studies, 06.12.2019 20:31



Biology, 06.12.2019 20:31


History, 06.12.2019 20:31


History, 06.12.2019 20:31

English, 06.12.2019 20:31


Mathematics, 06.12.2019 20:31

Advanced Placement (AP), 06.12.2019 20:31




Health, 06.12.2019 20:31