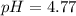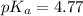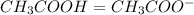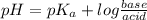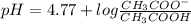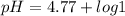Calculating the expected pH of the buffer solution: Given that the pKa for Acetic Acid is 4.77, calculate the expected pH of the buffer solutions using the Henderson-Hasselbalch equation and the concentrations of Acetic Acid and Acetate added to the 250 ml Erlenmeyer flask: pH

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:10
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

Chemistry, 23.06.2019 01:00
Animals that reproduce sexually either do it through external or internal fertilization. read the following statement and decide if it is true or false. birds reproduce through external reproduction which is because the female will then be able to protect the egg.
Answers: 1

Chemistry, 23.06.2019 01:10
A5.00 g of a in . g of at aa 5.00 g of b in . g of .?at .
Answers: 1
You know the right answer?
Calculating the expected pH of the buffer solution: Given that the pKa for Acetic Acid is 4.77, calc...
Questions

Biology, 27.08.2019 11:00


Mathematics, 27.08.2019 11:00

Chemistry, 27.08.2019 11:00



History, 27.08.2019 11:00

Chemistry, 27.08.2019 11:00


Mathematics, 27.08.2019 11:00

History, 27.08.2019 11:00

Mathematics, 27.08.2019 11:00

History, 27.08.2019 11:00


Mathematics, 27.08.2019 11:00



Mathematics, 27.08.2019 11:00