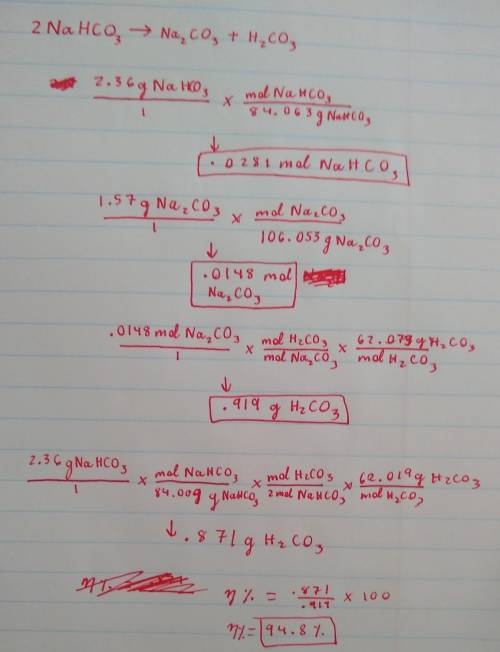ASAP
A 2.36-gram sample of NaHCO3 was completely decomposed in an experiment.
2NaHCO→ Na...

Chemistry, 24.07.2021 01:00 crookdamian21
ASAP
A 2.36-gram sample of NaHCO3 was completely decomposed in an experiment.
2NaHCO→ Na2CO3 + H2CO3
In this experiment, carbon dioxide and water vapors combine to form H2CO3. After decomposition, the Na2CO3 had a mass of 1.57 grams.
Determine the mass of the H2CO3 produced.
Calculate the percentage yield of H2CO3 for the reaction. Show your work or describe the calculation process in detail.
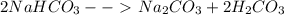

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
What type of electromagnetic radiation has a shorter wavelength than blue light
Answers: 2

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2

Chemistry, 23.06.2019 01:10
A5.00 g of a in . g of at aa 5.00 g of b in . g of .?at .
Answers: 1

Chemistry, 23.06.2019 04:31
Areaction is first order. if the initial reactant concentration is 0.0200 m, and 25.0 days later the concentration is 6.25 x 10-4 m, then its half-life is:
Answers: 1
You know the right answer?
Questions

SAT, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01


Mathematics, 02.10.2020 09:01

History, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01



Mathematics, 02.10.2020 09:01



History, 02.10.2020 09:01




Chemistry, 02.10.2020 09:01