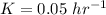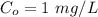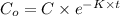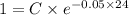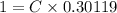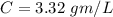Chemistry, 23.07.2021 02:20 Nathaliasmiles
A researcher is attempting to produce ethanol using an enzyme catalyzed batch reactor. The ethanol is produced from corn starch by first-order kinetics with a rate constant of 0.05 hr-1. Assuming the concentration of ethanol initially is 1 mg/L, what will be the concentration of ethanol (in mg/L) after 24 hours

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2


Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3

Chemistry, 23.06.2019 07:00
How does science use models to gain a better understanding of concepts?
Answers: 1
You know the right answer?
A researcher is attempting to produce ethanol using an enzyme catalyzed batch reactor. The ethanol i...
Questions








History, 29.11.2021 22:00


Physics, 29.11.2021 22:00


Spanish, 29.11.2021 22:00

Mathematics, 29.11.2021 22:00



English, 29.11.2021 22:00


Mathematics, 29.11.2021 22:00

Computers and Technology, 29.11.2021 22:00