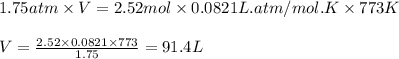Chemistry, 21.07.2021 20:00 imstressed
What volume of water is produced when 38.5 g of ethanol reacts with oxygen at 500°C at 1.75 atm? CH3CH2OH(g) + 3 O2(g)→ 2 CO2(g) + 3 H2O(g)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 23.06.2019 00:30
Nuclear decay is the spontaneous decay of one element into a. an x-ray b. a ray of light c. another element
Answers: 1
You know the right answer?
What volume of water is produced when 38.5 g of ethanol reacts with oxygen at 500°C at 1.75 atm?
CH...
Questions

History, 28.01.2020 05:31

Chemistry, 28.01.2020 05:31

Mathematics, 28.01.2020 05:31

Chemistry, 28.01.2020 05:31


Chemistry, 28.01.2020 05:31




Social Studies, 28.01.2020 05:31

Mathematics, 28.01.2020 05:31




Mathematics, 28.01.2020 05:31


Biology, 28.01.2020 05:31



Biology, 28.01.2020 05:31

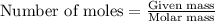 ......(1)
......(1)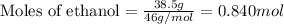
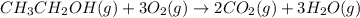
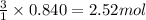 of water
of water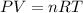 .......(2)
.......(2)![500^oC=[500+273]K=773K](/tpl/images/1397/4323/8da9a.png)