
Chemistry, 20.07.2021 01:30 rleiphart1
At 445oC, Kc for the following reaction is 0.020. 2 HI(g) <--> H2 (g) + I2 (g) A mixture of H2, I2, and HI in a vessel at 445oC has the following concentrations: [HI] = 1.5 M, [H2] = 2.50 M and [I2] = 0.05 M. Which one of the following statements concerning the reaction quotient, Qc, is TRUE for the above system?
a. Qc = Kc; the system is at equilibrium.
b. Qc is less than Kc; more H2 and I2 will be produced.
c. Qc is less than Kc; more HI will be produced.
d. Qc is greater than Kc; more HI will be produced.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be
Answers: 1

You know the right answer?
At 445oC, Kc for the following reaction is 0.020. 2 HI(g) <--> H2 (g) + I2 (g) A mixture of H2...
Questions


Social Studies, 26.01.2021 01:00


Geography, 26.01.2021 01:00



Mathematics, 26.01.2021 01:00






Mathematics, 26.01.2021 01:00

Arts, 26.01.2021 01:00



Biology, 26.01.2021 01:00

History, 26.01.2021 01:00

Mathematics, 26.01.2021 01:00

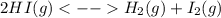
![Qc=\frac{[H_2][I_2]}{[HI]^2} \\Qc=(2.50Mx0.05M)/(1.5M)^2\\Qc=0.055](/tpl/images/1396/5648/39582.png)


