
Chemistry, 20.07.2021 01:00 alfarodougoy8lvt
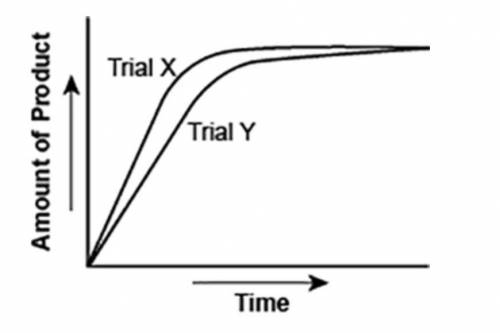
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different concentration of reactant was used during each trial, whereas the other factors were kept constant.
A graph is shown with two graph lines sloping upwards. The title on the x axis is Time and the title on the y axis is Amount of Product. The graph line with a steeper slope is labeled Trial X and the other is labeled Trial Y.
Which of the following statements explains which trial has a lower concentration of the reactant? (5 points)
Trial X, because the final volume of product formed is lower than Trial Y.
Trial X, because this reaction was initially fast and later stopped completely.
Trial Y, because the reaction was initially slow and later stopped completely.
Trial Y, because the volume of product formed per unit time is lower than Trial X.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1


Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1

Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2
You know the right answer?
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different...
Questions

Mathematics, 18.03.2021 01:10

History, 18.03.2021 01:10






Arts, 18.03.2021 01:10


History, 18.03.2021 01:10

Physics, 18.03.2021 01:10



Social Studies, 18.03.2021 01:10

Mathematics, 18.03.2021 01:10



Mathematics, 18.03.2021 01:10




