
Chemistry, 15.07.2021 21:50 avahrhey24
In aqueous solution the Ni2" ion forms a complex with four ammonia molecules. Write the formation constant expression for the equilibrium between the hydrated metal ion and the aqueous complex. Under that, write the balanced chemical equation for the first step in the formation of the complex K,=.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 21:20
The organs inside the body and how they function together
Answers: 3

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1
You know the right answer?
In aqueous solution the Ni2" ion forms a complex with four ammonia molecules. Write the formation co...
Questions

English, 06.12.2019 23:31

History, 06.12.2019 23:31

Mathematics, 06.12.2019 23:31





History, 06.12.2019 23:31

Health, 06.12.2019 23:31

Mathematics, 06.12.2019 23:31


Social Studies, 06.12.2019 23:31

Mathematics, 06.12.2019 23:31

Mathematics, 06.12.2019 23:31

English, 06.12.2019 23:31




Mathematics, 06.12.2019 23:31

![\frac{[Ni(H_2O)_3 (NH_3)]^{2+}}{[Ni(H_2O)_4]^{2+} [NH_3]}](/tpl/images/1394/7943/a809a.png) ".
".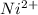 exist as
exist as ![[Ni(H_2O)_4]^{2+}](/tpl/images/1394/7943/49a05.png)
![[Ni(H_2O)_4]^{2+} + 4NH_3 \rightleftharpoons [Ni(NH_3)_4]^{2+} + H_2O](/tpl/images/1394/7943/ab9b4.png)
![K_f = \frac{[Ni(NH_3)_4]^{2+}}{[Ni(H_2O)_4^{2+}] [NH_3]^4}](/tpl/images/1394/7943/d666b.png)
![[H_2O]](/tpl/images/1394/7943/b5d70.png) as concentration of water will be const.
as concentration of water will be const.![[Ni(NH_3)_4]^{2+}](/tpl/images/1394/7943/dff73.png) proceeds via several steps,
proceeds via several steps,![[Ni(H_2O)_4]^{2+}+NH_3 \rightleftharpoons [Ni(H_2O)_3 (NH_3)]^{2+} + H_2O](/tpl/images/1394/7943/b2d29.png)
![K_1 = \frac{[Ni(H_2O)_3 (NH_3)]^{2+}}{[Ni(H_2O)_4]^{2+} [NH_3]}](/tpl/images/1394/7943/01675.png)


