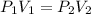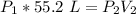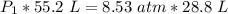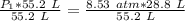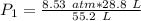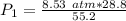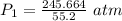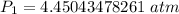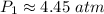Chemistry, 14.07.2021 01:20 cpcoolestkid4
A container is filled to a volume of 55.2 L at 61 °C. While keeping the
temperature constant, the volume is reduced to 28.8 L and the pressure at
the end was recorded to be 8.53 atm. What was the initial pressure inside
the container, in units of atm?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 16:00
How does blood clotting prevent the entry of pathogens through cuts and wounds? answer asap,, this is due tomorrow. will mark as brainliest or whatever you call it : )
Answers: 2

Chemistry, 22.06.2019 23:50
Which scientists contributed to the determination of how cfcs in clouds in the upper atmosphere could destroy ozone molecules
Answers: 1

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
You know the right answer?
A container is filled to a volume of 55.2 L at 61 °C. While keeping the
temperature constant, the v...
Questions

Chemistry, 19.10.2019 17:00

Mathematics, 19.10.2019 17:00

English, 19.10.2019 17:00


Mathematics, 19.10.2019 17:00




History, 19.10.2019 17:00

Social Studies, 19.10.2019 17:00






Mathematics, 19.10.2019 17:00



Mathematics, 19.10.2019 17:00

English, 19.10.2019 17:00