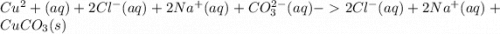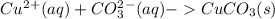Chemistry, 10.07.2021 02:30 stgitskaysie9028
g When copper(II) chloride and sodium carbonate solutions are combined, solid copper(II) carbonate precipitates, leaving a solution of sodium chloride. Write the conventional equation, total ionic equation, and net ionic equation for this reaction. (Use the lowest possible coefficients. Be sure to specify states such as (aq) or (s). If a box is not needed, leave it blank.)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 2

Chemistry, 22.06.2019 23:50
Which scientists contributed to the determination of how cfcs in clouds in the upper atmosphere could destroy ozone molecules
Answers: 1

Chemistry, 23.06.2019 07:00
Why do the strengths of london (dispersion) forces generally increase with increasing molecular size? choose one: a. heavier atoms have stronger attractions for each other than lighter atoms. b. dispersion forces are all equal in magnitude; there is no size dependence. c. dispersion forces arise from the attraction between the nuclei of atoms, and larger molecules have larger nuclei. d. dispersion forces arise from dipoles caused by the electron distribution being distorted. larger molecules have more electrons and, therefore, more distortions and a bigger force. e. dispersion forces depend on distance. larger molecules are farther apart and so the forces are smaller.
Answers: 2

You know the right answer?
g When copper(II) chloride and sodium carbonate solutions are combined, solid copper(II) carbonate p...
Questions


Mathematics, 21.09.2019 22:10

Physics, 21.09.2019 22:10



Mathematics, 21.09.2019 22:10

History, 21.09.2019 22:10

History, 21.09.2019 22:10


Mathematics, 21.09.2019 22:10

Mathematics, 21.09.2019 22:10





Social Studies, 21.09.2019 22:10


Spanish, 21.09.2019 22:10

Biology, 21.09.2019 22:10

Mathematics, 21.09.2019 22:10