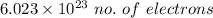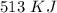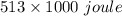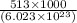Chemistry, 09.07.2021 22:10 kayadaniels2
it takes 513 kj to remove a mole of electrons from the atoms at the surface of a piece of metal. how much energy does it take to remove a single electron from n atom at the surface of the metal

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1

Chemistry, 22.06.2019 19:30
Astring vibrates with a frequency of 10 hz. why can't a person hear the sound waves produced by the vibrating string, no matter how large the amplitude of the waves? out! this is homework and due tomorrow! you so much!
Answers: 2

Chemistry, 23.06.2019 01:30
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1

Chemistry, 23.06.2019 09:30
How many moles of na2s2o3 are needed to react with 0.12mol of cl2? show work.
Answers: 1
You know the right answer?
it takes 513 kj to remove a mole of electrons from the atoms at the surface of a piece of metal. how...
Questions






Social Studies, 27.08.2019 01:30







Mathematics, 27.08.2019 01:40




Mathematics, 27.08.2019 01:40


Mathematics, 27.08.2019 01:40

Geography, 27.08.2019 01:40

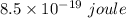 ".
".