

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:10
Seawater contains approximately 3.5%nacl by mass and has a density of 1.02 g/ml. what volume of seawater contains 7.5 g of sodium?
Answers: 2

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1

Chemistry, 22.06.2019 10:00
Main expenses you plan on making payments on a new car too. you want to spend 15% of your monthly net pay on the car payment, insurance, registration, and taxes combined. what is your monthly car allowance? $149.46 $298.91 $448.37 $597.83
Answers: 3

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2
You know the right answer?
4.If 15.00 mL of 3.00 M potassium iodide is needed to reach the equivalence point with 10.00 mL of l...
Questions




Mathematics, 21.04.2020 23:47

Mathematics, 21.04.2020 23:47

Mathematics, 21.04.2020 23:48

English, 21.04.2020 23:48



Chemistry, 21.04.2020 23:48

Spanish, 21.04.2020 23:48

Mathematics, 21.04.2020 23:48








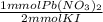 = 22.5 mmol Pb(NO₂)₃
= 22.5 mmol Pb(NO₂)₃

