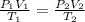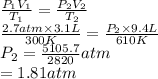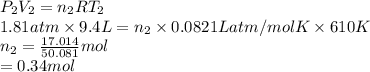A cylinder contains 3.1 L of oxygen at 300 K and 2.7 atm. The gas is heated, causing a piston in the cylinder to move outward. The heating causes the temperature to rise to 610 K and the volume of the cylinder to increase to 9.4 L.
How many moles of gas are in the cylinder?
Express your answer using two significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3
You know the right answer?
A cylinder contains 3.1 L of oxygen at 300 K and 2.7 atm. The gas is heated, causing a piston in the...
Questions

Biology, 10.10.2019 00:30


Mathematics, 10.10.2019 00:30

Business, 10.10.2019 00:30



Chemistry, 10.10.2019 00:30

Biology, 10.10.2019 00:30

Mathematics, 10.10.2019 00:30

Chemistry, 10.10.2019 00:30

Mathematics, 10.10.2019 00:30

English, 10.10.2019 00:30

Mathematics, 10.10.2019 00:30

History, 10.10.2019 00:30



History, 10.10.2019 00:30

Biology, 10.10.2019 00:30

History, 10.10.2019 00:30

Mathematics, 10.10.2019 00:30

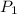 = 2.7 atm,
= 2.7 atm, 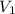 = 3.1 L,
= 3.1 L, 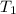 = 300 K
= 300 K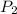 = ?,
= ?, 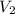 = 9.4 L,
= 9.4 L, 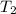 = 610 K
= 610 K