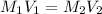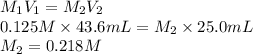Chemistry, 06.07.2021 21:30 ethangorrell67
What is the molarity of an HCl solution if 43.6 mL of a 0.125 M NaOH solution are needed to titrate a 25.0 mL sample of the acid according to the equation below (show your calculations)? NaOH(aq) + HCl(aq) → H2O(l) + NaCl(aq)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1

Chemistry, 22.06.2019 09:10
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 22.06.2019 23:00
What is the solubility-product constant of barium sulfate, baso4, if a saturated solution is 1.03 ´ 10-5 m?
Answers: 3
You know the right answer?
What is the molarity of an HCl solution if 43.6 mL of a 0.125 M NaOH solution are needed to titrate...
Questions

Mathematics, 20.09.2020 17:01








Mathematics, 20.09.2020 17:01


Mathematics, 20.09.2020 17:01

Arts, 20.09.2020 17:01

Biology, 20.09.2020 17:01

Advanced Placement (AP), 20.09.2020 17:01



Mathematics, 20.09.2020 17:01

Mathematics, 20.09.2020 17:01

Health, 20.09.2020 17:01

History, 20.09.2020 17:01

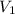 = 43.6 mL,
= 43.6 mL, 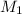 = 0.125 M
= 0.125 M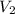 = 25.0 mL,
= 25.0 mL, 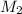 = ?
= ?