
Chemistry, 03.07.2021 04:30 sustaitaj2022
The normal boiling point of ethanol is 78.4 oC. Its enthalpy of vaporization is 38.6 kJ/mol. Estimate the vapor pressure of ethanol at 26.3 oC.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 08:00
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2
You know the right answer?
The normal boiling point of ethanol is 78.4 oC. Its enthalpy of vaporization is 38.6 kJ/mol. Estimat...
Questions

History, 01.06.2021 17:00

Social Studies, 01.06.2021 17:00


Mathematics, 01.06.2021 17:00



Mathematics, 01.06.2021 17:00

Chemistry, 01.06.2021 17:00


Mathematics, 01.06.2021 17:00

Mathematics, 01.06.2021 17:00

Mathematics, 01.06.2021 17:00





Mathematics, 01.06.2021 17:00

Mathematics, 01.06.2021 17:00

English, 01.06.2021 17:00

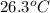 is 238.3 torr.
is 238.3 torr.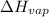 = 38.6 kJ/mol
= 38.6 kJ/mol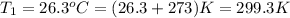
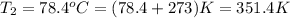
![ln\frac{P_{2}}{P_{1}} = \frac{\Delta H_{vap}}{R} [\frac{1}{T_{1}} - \frac{1}{T_{2}}]\\](/tpl/images/1388/5959/3b956.png)
![ln\frac{P_{2}}{P_{1}} = \frac{\Delta H_{vap}}{R} [\frac{1}{T_{1}} - \frac{1}{T_{2}}]\\ \\ln \frac{760 torr}{P_{1}} = \frac{38600 J}{8.314 J/mol K}[\frac{1}{299.3} - \frac{1}{351.4}]\\\frac{760}{P_{1}} = 3.18\\P_{1} = 238.3 torr](/tpl/images/1388/5959/42009.png)


