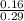Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
If a planet rotates 360 degrees during a 24 hour time period, what does that tell us about the planet? a. the middle of the planet is in darkness b. the seasons on the planet vary every day. c. the planet runs on a 12-hour time clock. d. the temperature on the planet varies daily.
Answers: 1

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 19:40
What causes different colors to appear in the sky? the absorption of light by air molecules the reflection of light by bodies of water the greenhouse effect in earth's atmosphere the scattering and reflection of light by dust particles
Answers: 2

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2
You know the right answer?
An aqueous solution contains 0.29 M of benzoic acid (HA) and 0.16 M of sodium benzoate (A-). If the...
Questions

Mathematics, 01.03.2021 16:50


Mathematics, 01.03.2021 16:50

History, 01.03.2021 16:50

Physics, 01.03.2021 16:50





Chemistry, 01.03.2021 16:50

Mathematics, 01.03.2021 16:50



Mathematics, 01.03.2021 16:50

Arts, 01.03.2021 16:50


English, 01.03.2021 16:50

Social Studies, 01.03.2021 16:50

Chemistry, 01.03.2021 16:50

![\frac{[A^-]}{[HA]}](/tpl/images/1388/3192/1d212.png)