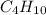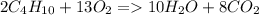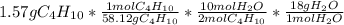Chemistry, 02.07.2021 04:10 danielbamf1414
Calculate the mass of water produced when 1.57g of butane reacts with excess oxygen

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:00
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1

Chemistry, 22.06.2019 05:30
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
You know the right answer?
Calculate the mass of water produced when 1.57g of butane reacts with excess oxygen...
Questions

Mathematics, 18.10.2020 17:01

Mathematics, 18.10.2020 17:01





Health, 18.10.2020 17:01

Mathematics, 18.10.2020 17:01

History, 18.10.2020 17:01


Mathematics, 18.10.2020 17:01

Advanced Placement (AP), 18.10.2020 17:01

Arts, 18.10.2020 17:01

Mathematics, 18.10.2020 17:01

Mathematics, 18.10.2020 17:01

Computers and Technology, 18.10.2020 17:01

Physics, 18.10.2020 17:01

Geography, 18.10.2020 17:01

Computers and Technology, 18.10.2020 17:01

Computers and Technology, 18.10.2020 17:01