
Chemistry, 02.07.2021 02:50 beccaxhope
35 POINTS - WILL GIVE BRAINLIEST!
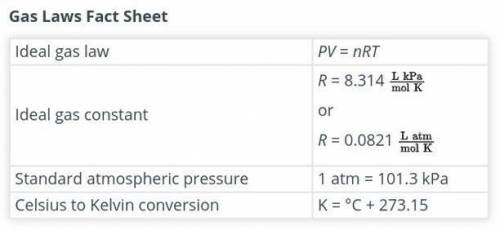
One of the main components of an airbag is the gas that fills it. As part of the design process, you need to
determine the exact amount of nitrogen that should be produced. Calculate the number of moles of
nitrogen required to fill the airbag. Show your work. Assume that the nitrogen produced by the chemical
reaction is at a temperature of 495°C and that nitrogen gas behaves like an ideal gas. Use this fact sheet
e to review the ideal gas law.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1
You know the right answer?
35 POINTS - WILL GIVE BRAINLIEST!
One of the main components of an airbag is the gas that fills it....
Questions



Biology, 09.11.2020 20:10

Mathematics, 09.11.2020 20:10




Geography, 09.11.2020 20:10

Mathematics, 09.11.2020 20:10





Biology, 09.11.2020 20:10

Mathematics, 09.11.2020 20:10


Social Studies, 09.11.2020 20:10

Mathematics, 09.11.2020 20:10




