Chemistry, 29.06.2021 02:10 fjjjjczar8890
Calculate the number of molecules of carbon (IV) oxide
produced when 10g of CaCO3 is treated with 100cm of
0.2mol dm HCl. The equation of the reaction is
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(1) + CO2(g)
ICa=40 (=12 O=161

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

You know the right answer?
Calculate the number of molecules of carbon (IV) oxide
produced when 10g of CaCO3 is treated with 1...
Questions



Health, 13.09.2019 17:30


Social Studies, 13.09.2019 17:30


Mathematics, 13.09.2019 17:30

Mathematics, 13.09.2019 17:30

Mathematics, 13.09.2019 17:30



Mathematics, 13.09.2019 17:30








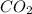 produced is 0.44 g
produced is 0.44 g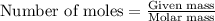 ......(1)
......(1)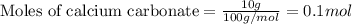
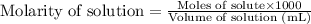 .....(2)
.....(2)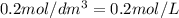 (Conversion factor:
(Conversion factor: 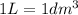
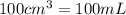 (Conversion factor:
(Conversion factor: 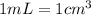
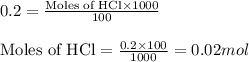
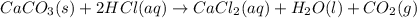
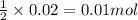 of calcium carbonate
of calcium carbonate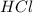 produces 1 mole of
produces 1 mole of