Calculate [H3O+] and [OH−] for each of the following solutions at 25 ∘C given the pH.
pH= 2.89...

Chemistry, 28.06.2021 20:50 marlenemedina247
Calculate [H3O+] and [OH−] for each of the following solutions at 25 ∘C given the pH.
pH= 2.89

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

You know the right answer?
Questions





Mathematics, 22.04.2020 16:46





English, 22.04.2020 16:46



English, 22.04.2020 16:46


Biology, 22.04.2020 16:46



Mathematics, 22.04.2020 16:46

Biology, 22.04.2020 16:47

Biology, 22.04.2020 16:47

![[H_{3}O^{+}]](/tpl/images/1385/4271/71b58.png) is 0.0012 M and
is 0.0012 M and ![[OH^{-}]](/tpl/images/1385/4271/e46dd.png) is
is 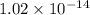 .
. ![pH = - log [H^{+}]\\2.89 = - log [H^{+}]\\conc. H^{+} = 0.0012 M](/tpl/images/1385/4271/e5499.png)
![pOH = - log [OH^{-}]\\13.99 = - log [OH^{-}]\\conc. OH^{-} = 1.02 \times 10^{-14} M](/tpl/images/1385/4271/f05e1.png)


