Chemistry, 28.06.2021 15:10 robclark128
At 35°C, K = 1.6 × 10^-5 for the reaction
2 NOCl(g) ⇌ 2 NO(g) + Cl2(g)
Calculate the concentrations of all species at equilibrium for each of the following original mixtures.
a. 2.0 mol pure NOCl in a 2.0 L flask
b. 2.0 mol NOCl and 1.0 mol Cl2 in a 1.0 L flask

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the nature of the ca-cl bond in a molecule of calcium chloride (cacl2) if the electronegativity value of calcium is 1.0 and that of chlorine is 3.16?
Answers: 1

Chemistry, 21.06.2019 18:40
What kind of ion is contained in salts that produce an acidic solution? a positive ion that attracts a proton from water a positive ion that releases a proton to water a negative ion that attracts a proton from water a negative ion that releases a proton to water
Answers: 1

Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2

Chemistry, 22.06.2019 10:30
Which characteristics can be used to differentiate star systems? check all that apply.
Answers: 2
You know the right answer?
At 35°C, K = 1.6 × 10^-5 for the reaction
2 NOCl(g) ⇌ 2 NO(g) + Cl2(g)
Calculate the co...
Calculate the co...
Questions



Mathematics, 23.12.2019 05:31

Biology, 23.12.2019 05:31




Social Studies, 23.12.2019 05:31



Social Studies, 23.12.2019 05:31


World Languages, 23.12.2019 05:31

History, 23.12.2019 05:31

Chemistry, 23.12.2019 05:31



Chemistry, 23.12.2019 05:31

English, 23.12.2019 05:31

Biology, 23.12.2019 05:31

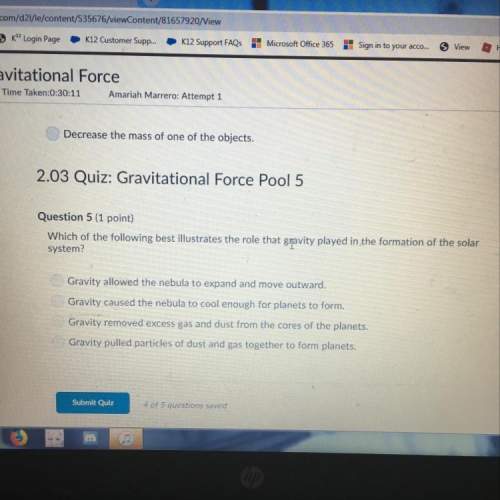
 = 4.0*10^-3
= 4.0*10^-3