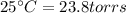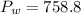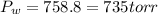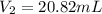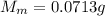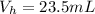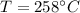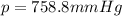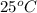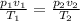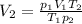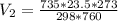4. A piece of metal weighing 0.0713 g was placed in a eudiometer containing dilute aqueous HCl. After the metal fully dissolved, 23.5 mL of hydrogen gas was collected by displace-ment of water and a 400 mm column of water was observed. The water temperature was 258C and the barometric pressure was 758.8 mm Hg (torr). Refer to the Introduction and data sheet to solve the following problems. a) What is the vapor pressure of the water vapor in the column? (Consult Appendix E.)

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 22:30
Is the idea of spontaneous generation supported by redi's experiment? justify your answer in 2-3 sentences?
Answers: 1

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1
You know the right answer?
4. A piece of metal weighing 0.0713 g was placed in a eudiometer containing dilute aqueous HCl. Afte...
Questions

English, 19.07.2019 06:50

English, 19.07.2019 06:50

Mathematics, 19.07.2019 06:50


Biology, 19.07.2019 06:50



Mathematics, 19.07.2019 06:50


Mathematics, 19.07.2019 06:50



Mathematics, 19.07.2019 06:50

Health, 19.07.2019 06:50


Mathematics, 19.07.2019 06:50

History, 19.07.2019 06:50

History, 19.07.2019 06:50


Mathematics, 19.07.2019 06:50