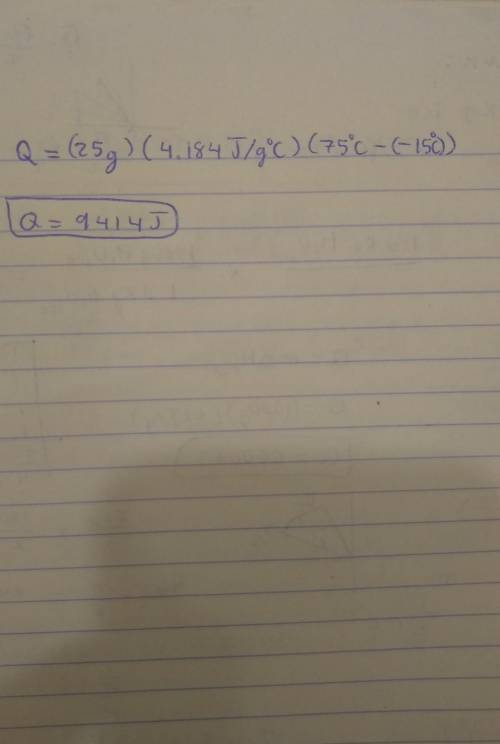Chemistry, 24.06.2021 03:30 sevaramirabell
Note: Please show all work and calculation setups to get full credit. T. he following may be used on this assignment: specific heat of (water=4.184 J/g oC; ice=2.03 J/g oC; steam=1.99 184 J/g oC); heat of fusion of water=80. cal/g; heat of vaporization=540 cal/g; 1cal=4.184J. Calculate the energy required (in J) to convert 25 g of ice at -15 oC to water at 75 oC.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 14:00
Ascientist measures the speed of sound in a monatomic gas to be 449 m/s at 20∘c. what is the molar mass of this gas?
Answers: 2

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

Chemistry, 23.06.2019 01:50
Drag the tiles to the correct locations. each tile can be used more than once, but not all tiles will be used. one or more locations will remain empty. nitrosyl fluoride has the chemical formula nof nitrogen has five valence electrons, oxygen has six, and fluorine has seven. complete the lewis structure for this covalent compound. f n = = = . : : 0 : reset next um. all rights reserved us 2
Answers: 2
You know the right answer?
Note: Please show all work and calculation setups to get full credit. T. he following may be used on...
Questions


Mathematics, 04.10.2021 23:30

Mathematics, 04.10.2021 23:40



Mathematics, 04.10.2021 23:40

Mathematics, 04.10.2021 23:40

Social Studies, 04.10.2021 23:40

Mathematics, 04.10.2021 23:40



Geography, 04.10.2021 23:40


Physics, 04.10.2021 23:40

Mathematics, 04.10.2021 23:40

SAT, 04.10.2021 23:40

Mathematics, 04.10.2021 23:40

Mathematics, 04.10.2021 23:40

Mathematics, 04.10.2021 23:40