Chemistry, 21.06.2021 20:50 kaylatunell123
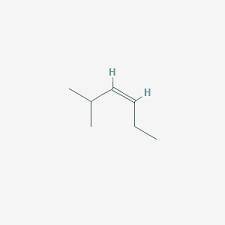
Compounds X and Y both have the formula C7H14. Both X and Y react with one molar equivalent of hydrogen in the presence of a palladium catalyst to form 2-methylhexane. The heat of hydrogenation of X is greater than that of Y. Both X and Y react with HCl to give the same single C7H15Cl compound as the major product. What is the structure of X?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1


Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3
You know the right answer?
Compounds X and Y both have the formula C7H14. Both X and Y react with one molar equivalent of hydro...
Questions

Mathematics, 30.05.2020 19:03


Physics, 30.05.2020 19:03





Mathematics, 30.05.2020 19:57

Arts, 30.05.2020 19:57

Mathematics, 30.05.2020 19:57

Mathematics, 30.05.2020 19:57




Mathematics, 30.05.2020 19:57



Mathematics, 30.05.2020 19:57


Mathematics, 30.05.2020 19:57