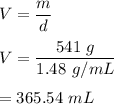Chemistry, 21.06.2021 05:20 sierram298
2) If the density of chloroform is 1.48 g/mL, what is the volume of 541 g of chloroform?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2


Chemistry, 23.06.2019 01:00
Which statement best describes isomers? a. isomers are alcohols that have the same functional group. b. isomers have at least one carbon-carbon double bond. c. isomers have the same molecular formula but different structural properties.
Answers: 1

Chemistry, 23.06.2019 01:30
Witch two conditions can limit the usefulness of the kinetic molecular theory in describing gas behavior?
Answers: 2
You know the right answer?
2) If the density of chloroform is 1.48 g/mL, what is the volume of 541 g of chloroform?...
Questions




History, 08.07.2019 10:30


Social Studies, 08.07.2019 10:30

Chemistry, 08.07.2019 10:30


History, 08.07.2019 10:30


History, 08.07.2019 10:30


Mathematics, 08.07.2019 10:30


Biology, 08.07.2019 10:30

Biology, 08.07.2019 10:30

Computers and Technology, 08.07.2019 10:30



Mathematics, 08.07.2019 10:30