
Chemistry, 19.06.2021 18:10 pattykline
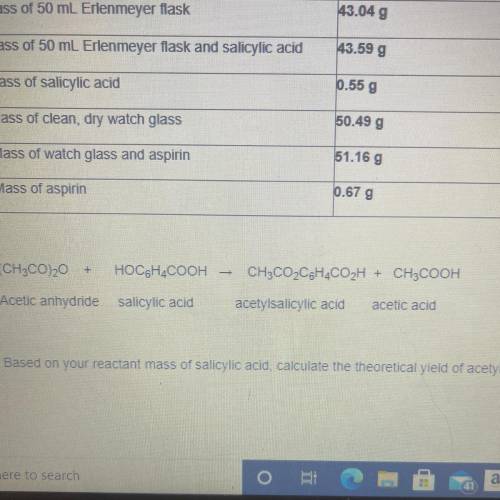
Based on your reactant mass of salicylic acid calculate the theoretical trio of acetysalicylic acid
The chart is in the picture
Please help I’ll give you brainiest

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1

Chemistry, 23.06.2019 08:00
What is the temperature in kelvin of a gas if it is allowed to expand from 1.50 l to 4.50 l? the initial temperature is 10.0°c and pressure is constant throughout the change. which equation should you use? t2= v2/v1 t1 what is the final temperature? ⇒ 849 k these are the answers.
Answers: 1

Chemistry, 23.06.2019 08:40
The half-life of a certain element is 100 days. how many half-lives will it be before only one eighth of this elementremains?
Answers: 1
You know the right answer?
Based on your reactant mass of salicylic acid calculate the theoretical trio of acetysalicylic acid...
Questions





Mathematics, 21.05.2021 17:10


Mathematics, 21.05.2021 17:10


Mathematics, 21.05.2021 17:10

Mathematics, 21.05.2021 17:10

Computers and Technology, 21.05.2021 17:10

Computers and Technology, 21.05.2021 17:10

Chemistry, 21.05.2021 17:10

Mathematics, 21.05.2021 17:10

World Languages, 21.05.2021 17:10

History, 21.05.2021 17:10

Mathematics, 21.05.2021 17:10





