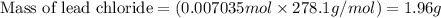Chemistry, 18.06.2021 23:50 tonydeanfbg8706
g When aqueous solutions of and are mixed, a solid forms. Determine the mass of solid formed when 140.7 mL of 0.1000 M is mixed with an excess of an aqueous solution of .

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:10
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2


Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2
You know the right answer?
g When aqueous solutions of and are mixed, a solid forms. Determine the mass of solid formed when 14...
Questions

Mathematics, 10.10.2021 14:00



Mathematics, 10.10.2021 14:00


Mathematics, 10.10.2021 14:00



Social Studies, 10.10.2021 14:00


Biology, 10.10.2021 14:00

Mathematics, 10.10.2021 14:00



Biology, 10.10.2021 14:00

Mathematics, 10.10.2021 14:00


Mathematics, 10.10.2021 14:00

English, 10.10.2021 14:00

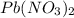 are mixed, a solid forms. Determine the mass of solid formed when 140.7 mL of 0.1000 M NaCl is mixed with an excess of an aqueous solution of
are mixed, a solid forms. Determine the mass of solid formed when 140.7 mL of 0.1000 M NaCl is mixed with an excess of an aqueous solution of  .....(1)
.....(1)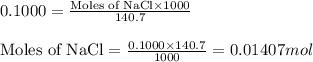
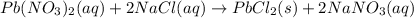
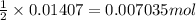 of lead chloride
of lead chloride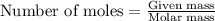 ......(2)
......(2)