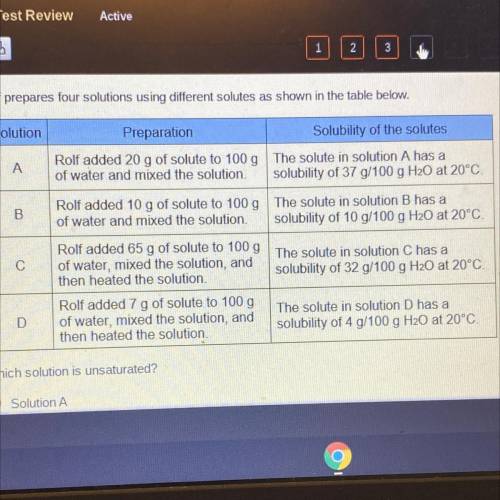
Rolf prepares four solutions using different solutes as shown in the table below.
Solution
Pr...

Rolf prepares four solutions using different solutes as shown in the table below.
Solution
Preparation
Solubility of the solutes
A
Rolf added 20 g of solute to 100 g The solute in solution A has a
of water and mixed the solution solubility of 37 g/100 g H20 at 20°C!
B
Rolf added 10 g of solute to 100 g The solute in solution B has a
of water and mixed the solution. solubility of 10 g/100 g H20 at 20°C.
С
Rolf added 65 g of solute to 100g The solute in solution C has a
of water, mixed the solution, and
then heated the solution,
solubility of 32 g/100 g H20 at 20°C
D
Rolf added 7 g of solute to 100 g
of water, mixed the solution, and
then heated the solution
The solute in solution D has a
solubility of 4 g/100 g H20 at 20°C.
Which solution is unsaturated?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which of the following statements is true about planck’s law
Answers: 1

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

You know the right answer?
Questions


Geography, 03.10.2020 01:01

English, 03.10.2020 01:01



Computers and Technology, 03.10.2020 01:01

History, 03.10.2020 01:01



Chemistry, 03.10.2020 01:01

English, 03.10.2020 01:01


History, 03.10.2020 01:01

Social Studies, 03.10.2020 01:01

English, 03.10.2020 01:01

Geography, 03.10.2020 01:01



Health, 03.10.2020 01:01



