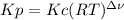Chemistry, 14.06.2021 17:00 therealrg10
Equilibrium constants for gases can be expressed in terms of concentrations, Kc, or in terms of partial pressures, Kp. Which one of the following statements regarding Kc and Kp is correct?
a. Kc and Kp are equal when all stoichiometric coefficients in the balanced reaction equation equal one.
b. Kc and Kp are equal when the conditions are standard (P= 1 atm, T=298 K)
c. Kc and Kp are equal when the sum of the stoichiometric coefficients for the products equals the sum of the stoichiometric coefficients for the reactants.
d. Kc and Kp can never be equal.
e. Kc and Kp have the same values but different units.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Acylinder is filled with 2.00 moles of nitrogen, 3.00 moles of argon and 5.00 moles of helium. if the gas mixture is at stp, what is the partial pressure of the argon
Answers: 1

Chemistry, 22.06.2019 08:00
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1

Chemistry, 22.06.2019 20:30
Select all the correct answers.which compounds have the empirical formula ch20? (multiple answers)a.c2h4o2b.c3h603c.ch2o2d.c5h1005e.c6h1206
Answers: 2

You know the right answer?
Equilibrium constants for gases can be expressed in terms of concentrations, Kc, or in terms of part...
Questions

Mathematics, 14.07.2020 01:01

Mathematics, 14.07.2020 01:01

Mathematics, 14.07.2020 01:01



Mathematics, 14.07.2020 01:01



Mathematics, 14.07.2020 01:01



Computers and Technology, 14.07.2020 01:01

Social Studies, 14.07.2020 01:01

English, 14.07.2020 01:01



Mathematics, 14.07.2020 01:01


English, 14.07.2020 01:01

Health, 14.07.2020 01:01