Molybdenum (Mo) crystallizes with a body-centered cubic lattice and has an atomic radius of 136.1 pm.
a. Calculate the density of molybdenum in g/cm^3. Show all work. Include units with all numbers and show how units cancel.
b. Calculate the density of molybdenum in g/cm^3

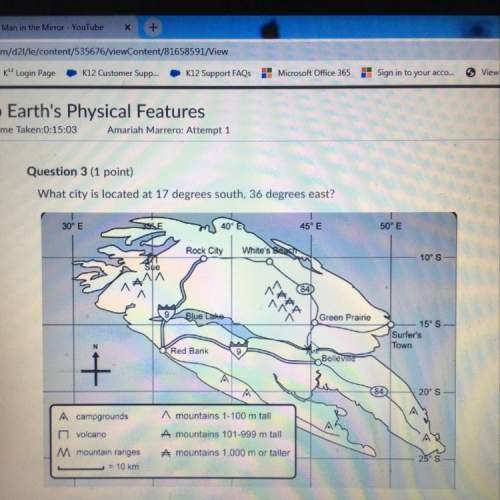
Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which statement about sound is not true? a. air particles travel with sound waves. b. sound waves cannot travel through a vacuum. c. sound waves exist even if no one hears them. d. air particles vibrate along the path of a sound wave.
Answers: 1

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2

Chemistry, 22.06.2019 08:00
Will give ! what are the advantages and disadvantages of nuclear power? check all that apply. one advantage of nuclear energy is that it does not produce carbon dioxide emissions. storage of nuclear waste is a short-term problem associated with nuclear energy. the problem with uranium mining is that a large quantity of uranium must be extracted to meet energy needs because the energy release from uranium fission is so low. safe operation of a nuclear power plant can be jeopardized by a human mistake.
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2
You know the right answer?
Molybdenum (Mo) crystallizes with a body-centered cubic lattice and has an atomic radius of 136.1 pm...
Questions

Mathematics, 29.02.2020 01:14

Mathematics, 29.02.2020 01:14




English, 29.02.2020 01:14

Advanced Placement (AP), 29.02.2020 01:14


Computers and Technology, 29.02.2020 01:14




Mathematics, 29.02.2020 01:14

English, 29.02.2020 01:14

Mathematics, 29.02.2020 01:14