
Chemistry, 12.06.2021 01:40 benjaminmccutch
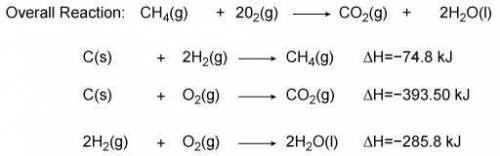
Use the information from the diagram to calculate the enthalpy of combustion for methane.
Question 2 options:
A)
+752 kJ
B)
–921 kJ
C)
–604 kJ
D)
+604 kJ

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 08:50
If two atoms are bonded to a central atom with no lone pairs,how will they be arranged
Answers: 3

Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1

Chemistry, 22.06.2019 12:30
What is the percent composition of ca(oh)2? 37.7% ca, 53.0% o, and 10.3% h 45.5% ca, 38.2% o, and 16.3% h 54.0% ca, 43.0% o, and 2.7% h 64.7% ca, 27.0% o, and 8.3% h
Answers: 2
You know the right answer?
Use the information from the diagram to calculate the enthalpy of combustion for methane.
Question...
Questions





Mathematics, 21.12.2020 22:30

Social Studies, 21.12.2020 22:30

Mathematics, 21.12.2020 22:30


Mathematics, 21.12.2020 22:30


Chemistry, 21.12.2020 22:30


Mathematics, 21.12.2020 22:30




Mathematics, 21.12.2020 22:30

Mathematics, 21.12.2020 22:30

Business, 21.12.2020 22:30



